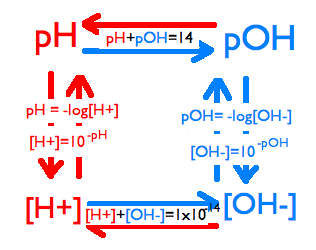
Solving for pH, pOH, [H+], [OH-]
In this section you will learn how to calculate for pH, pOH, [H+] and [OH-]
Before we begin, it is important that you remember what pH, pOH, [H+] and [OH-] mean.
pH: Potential of Hydrogen
pOH: Potential of Hydroxide
[H+]: Hydrogen Ions
[OH-]: Hydroxide Ions
If you want more information, you can click here.
These are the formulas for pH, pOH, [H+] and [OH-]:
pH: Potential of Hydrogen
pOH: Potential of Hydroxide
[H+]: Hydrogen Ions
[OH-]: Hydroxide Ions
If you want more information, you can click here.
These are the formulas for pH, pOH, [H+] and [OH-]:
pH= -log[H+]
pOH= -log[OH-]
[H+]= 2nd log (-pH)
[OH-]= 2nd log (-pOH)
pH+pOH= 14
pOH= -log[OH-]
[H+]= 2nd log (-pH)
[OH-]= 2nd log (-pOH)
pH+pOH= 14
***In case you don't know what "log" is, it can be found here. Additionally, 2nd log can be found be pressing "2nd" on the top of the calculator then pressing "log"***
Examples:
If a solution has a pH of 4, what is the [H+]?
Since we know the pH, we can use the formula [H+] = 2nd log (pH)
Plug in the known information: [H+] = 2nd log (-4)
Solve the equation on a graphing calculator: [H+] = .0001 or 1 E-4
If a solution has a pOH of 9, what is the [H+]?
Since we know the pOH, we can use the formula pH + pOH = 14
Plug in the known information: pH + 9 = 14
Solve the equation: pH = 5
We can then use another equation to help solve for [H+]: [H+] = 2nd log (-pH)
Plug in the known information: [H+] = 2nd log (-5)
Solve the equation on a graphing calculator: [H+] = .00001 or 1 E-5
If a solution has [OH-] = 3 E-3, what is the pH?
Since we know the [OH-], we can use the formula pOH = -log [OH-]
Plug in the known information: pOH = -log [3 E-3]
Solve the equation on a graphing calculator: pOH = 2.5
We can then use another equation to help solve for pH: pH + pOH = 14
Plug in the known information: 2.5 + pH = 14
Solve the equation: pH = 11.5
If a solution has a pH of 4, what is the [H+]?
Since we know the pH, we can use the formula [H+] = 2nd log (pH)
Plug in the known information: [H+] = 2nd log (-4)
Solve the equation on a graphing calculator: [H+] = .0001 or 1 E-4
If a solution has a pOH of 9, what is the [H+]?
Since we know the pOH, we can use the formula pH + pOH = 14
Plug in the known information: pH + 9 = 14
Solve the equation: pH = 5
We can then use another equation to help solve for [H+]: [H+] = 2nd log (-pH)
Plug in the known information: [H+] = 2nd log (-5)
Solve the equation on a graphing calculator: [H+] = .00001 or 1 E-5
If a solution has [OH-] = 3 E-3, what is the pH?
Since we know the [OH-], we can use the formula pOH = -log [OH-]
Plug in the known information: pOH = -log [3 E-3]
Solve the equation on a graphing calculator: pOH = 2.5
We can then use another equation to help solve for pH: pH + pOH = 14
Plug in the known information: 2.5 + pH = 14
Solve the equation: pH = 11.5