Conjugates
What is a Conjugate?
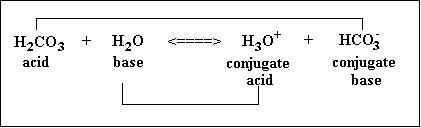
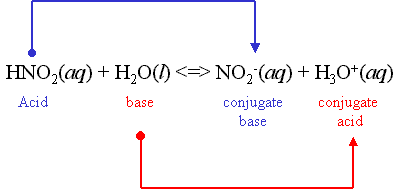
Conjugate Acid: Created when a base gains a H+ ion
Conjugate Base: Created when an acid gains a OH- ion
In a acid-base reaction, the following equation is used:
Acid + Base = Conjugate Acid + Conjugate Base
How to Identify Conjugate Acids and Bases:
Look below for two examples of acid and base reactions. It shows the reactants and how they become conjugate acids and bases as products.
Conjugate Base: Created when an acid gains a OH- ion
In a acid-base reaction, the following equation is used:
Acid + Base = Conjugate Acid + Conjugate Base
How to Identify Conjugate Acids and Bases:
- Identify the acid by locating the [H+] and the base by locating the [OH-]
- ***Hint! These will ALWAYS be on the left side of the equation as reactants of the reaction***
- Follow the proton to locate the conjugate acid and base of the reaction
- ***Hint! These will ALWAYS be on the right side of the equation as products of the reaction***
Look below for two examples of acid and base reactions. It shows the reactants and how they become conjugate acids and bases as products.